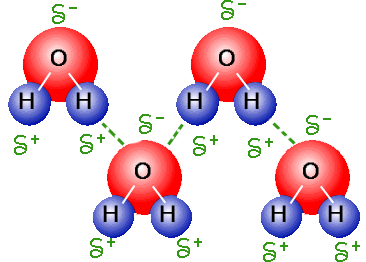
Hydrogen Bonding
A special type of Dipole-Dipole attractive force is Hydrogen Bonding. This type of bonding occurs when a hydrogen atom is bonded to an oxygen, nitrogen, or fluorine atom. This attraction between the two atoms is a dipole-dipole attraction, however, because of the large difference in electronegativity the bond also has some features of covalent bonding--it's directional, stronger than a van der waals interaction, and normally involves a limited number of interaction partners. This type of bonding leads to more intense physical properties for these molecules than for other molecules with Dipole-Dipole attractions. For example, water, a molecule with two hydrogen bonds, boils at 100 degrees Celsius while PH3 boils at -87.7 degrees Celsius. The difference in their boiling points is due to the two hydrogen bonds that water has as opposed to the normal Dipole-Dipole attraction force that phosphine experiences.
An example of hydrogen bonding between water molecules. These bonds are the reason why water has less density when it freezes.