Ionic Bonding
Ionic bonding exists between the lattice structures of ionic crystals. It is a chemical bond in which one atom cedes an electron to another, generating an electrical force that holds the atoms together. Essentially, the very charged regions of the ionic molecule interact with the oppositely charged regions on other molecules, forming a lattice structure. They are stronger than Dipole-Dipole attractions because of the change in electronegativity between the two elements is much greater.
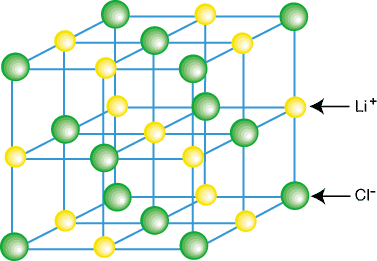
A crystal of Lithium Chloride (LiCl). Note how the cations are adjacent with four other anions and vice versa for the anions.