Critical Point: The temperature and pressure at which the liquid and gaseous phases of a pure and stable substance become identical.
Critical Pressure: The minimum applied pressure required at the critical temperature to liquefy a gas.
Critical Temperature: The temperature above which a gas cannot be liquefied, regardless of the pressure applied to it.
Dipole-Dipole Attraction: Molecules experiencing dipole moments that attract each other electrostatically by lining up so that the positive and negative ends are near each other.
Electronegativity: the relative strength of an atom in terms of its ability to draw electrons.
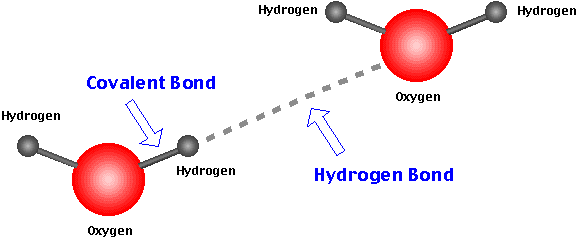
Hydrogen Bonding: A specific type of dipole-dipole force that is outstandingly strong and is normally observed among molecules in which hydrogen is bound to a highly electronegative atom such as nitrogen, oxygen, or fluorine.
Intermolecular forces: Forces that involve covalent or ionic bonding, and weaker interactions that occur between the molecules of a substance.
Ionic Forces: When a compound is made up of an ionic bond, it has ionic forces as its IMF.
London Dispersion Forces: Intermolecular forces that exist among the atoms of noble gases and nonpolar molecules.
Network Covalent: A compound where the atoms are covalently bonded in a network.
Nonpolar: when a molecule has an even distribution of electrons
Normal Boiling Point: The temperature at which a liquid boils at 1 atm.
Normal Melting Point: The temperature at which a solid melts at 1 atm.
Polar: describes a molecule which has an uneven distribution of electrons due to the differences in electronegativity of the atoms which comprise it.
Supercritical Fluid: Any substance past the critical point where the liquid and gasses phases are indistinguishable.
Surface Tension: The resistance of a liquid to an increase of its surface area; liquids with relatively large intermolecular forces (usually polar molecules) tend to have high surface tensions.
Triple Point: Temperature and pressure at which the solid, liquid, and gaseous phases of a given substance are all at equilibrium with one another.
Critical Pressure: The minimum applied pressure required at the critical temperature to liquefy a gas.
Critical Temperature: The temperature above which a gas cannot be liquefied, regardless of the pressure applied to it.
Dipole-Dipole Attraction: Molecules experiencing dipole moments that attract each other electrostatically by lining up so that the positive and negative ends are near each other.
Electronegativity: the relative strength of an atom in terms of its ability to draw electrons.
Hydrogen Bonding: A specific type of dipole-dipole force that is outstandingly strong and is normally observed among molecules in which hydrogen is bound to a highly electronegative atom such as nitrogen, oxygen, or fluorine.
Intermolecular forces: Forces that involve covalent or ionic bonding, and weaker interactions that occur between the molecules of a substance.
Ionic Forces: When a compound is made up of an ionic bond, it has ionic forces as its IMF.
London Dispersion Forces: Intermolecular forces that exist among the atoms of noble gases and nonpolar molecules.
Network Covalent: A compound where the atoms are covalently bonded in a network.
Nonpolar: when a molecule has an even distribution of electrons
Normal Boiling Point: The temperature at which a liquid boils at 1 atm.
Normal Melting Point: The temperature at which a solid melts at 1 atm.
Polar: describes a molecule which has an uneven distribution of electrons due to the differences in electronegativity of the atoms which comprise it.
Supercritical Fluid: Any substance past the critical point where the liquid and gasses phases are indistinguishable.
Surface Tension: The resistance of a liquid to an increase of its surface area; liquids with relatively large intermolecular forces (usually polar molecules) tend to have high surface tensions.
Triple Point: Temperature and pressure at which the solid, liquid, and gaseous phases of a given substance are all at equilibrium with one another.